 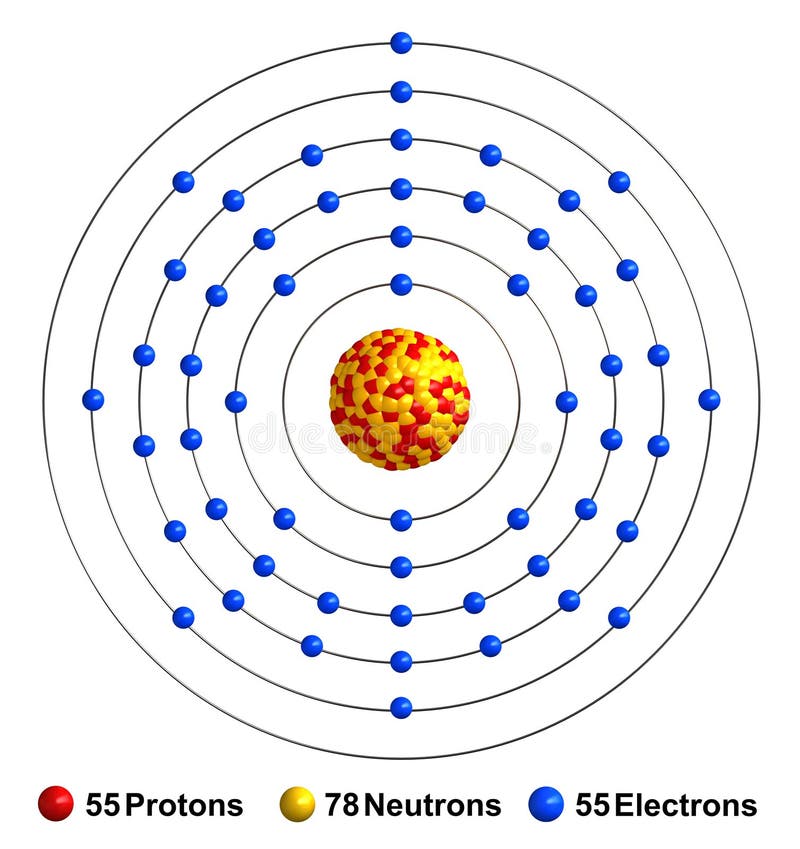
After the Caesium has been formed into a solid, it is ready for use in a variety of applications. This can be done via melting, casting, or any other appropriate technique. The next step is to form the Caesium into a solid.After it has been heated, it is ready to be formed into a solid. This is done to increase the reactivity of the Caesium, which is necessary for the production of certain compounds. Then, it is heated to a certain temperature.After the purification process is complete, it is ready for use. 
This involves distilling the source material to remove any impurities and creating a purer form of Caesium. Next, the source is put through a purification process.It is important to select a source that is free of impurities, as impurities can cause an adverse reaction when preparing it. It can be obtained either in elemental form or in a variety of compounds. First, the Caesium source is purchased.The following steps provide the optimal method of preparing Caesium. In order to obtain the highest quality Caesium, adequate preparation is essential. It is a key component of atomic clocks and an essential ingredient for the production of certain composite materials and alloys. Preparation of CaesiumĬaesium is an essential element for a variety of scientific, technological, and industrial areas of activity. It can also form complexes with other molecules, such as nitrogen, hydrogen and water, resulting in the formation of caesium nitrate, caesium hydroxide and caesium hydrate, respectively. The most stable form of caesium is the caesium chloride (CsCl) molecule, which consists of a single cesium atom surrounded by four chloride ions in a cube structure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
